April 30th, 2014 — The American Orthotic and Prosthetic Association (AOPA) is pleased to provide this statement, which was delivered to the House Ways & Means Subcommittee today, concerning Medicare fraud and the delivery of care to Medicare beneficiaries who have suffered a loss of a limb or impaired use of a limb or the spine. AOPA, founded in 1917, is the largest orthotic and prosthetic trade association with a national membership that draws from all segments of the field of artificial limbs and customized bracing for the benefit of patients who have experienced limb loss or limb impairment resulting from a chronic disease or health condition. Members include patient care facilities, manufacturers and distributors of prostheses, orthoses, and related products, plus educational and research institutions.
Annual Medicare spending for custom orthotics and all prosthetics is less than one percent of all Medicare spending. However, Medicare fraud has an outsized impact on the beneficiaries whose limb loss or impairment results in the need for orthotics or prosthetics. Patients treated by AOPA’s members already are confronted with the trauma of limb loss or impairment, loss of mobility, diminished independence, and sometimes financial hardship. When seen by a fraudulent supplier, the patient also oftentimes experiences a financial loss after paying for a device that is inappropriate or never delivered. Additionally, a patient in this situation has to find another supplier and make another copayment, and he or she may lose important time in the rehabilitation process. Dobson-DaVanzo’s research concluded that nearly one-thirdof the $3.62 billion CMS paid between 2007-2011 for orthotic and prosthetic services for Medicare beneficiaries went to unlicensed providers, as well as those who fail to meet the accreditation requirement legislated by Congress in 2000. Additional research by Dobson-DaVanzo tracking Medicare data has demonstrated the overall cost-effectiveness of O&P care. For example, the analytic work indicated that over the first eighteen months patients who receive spinal orthoses had total Medicare episode payments that were 0.3% lower than those who did not receive orthotic bracing for the comparable back ailment. These are important trends for saving Medicare dollars.
AOPA and its members believe the best way to fight fraud in the orthotics and prosthetics sector is to prevent fraud in the first place. We also believe that it is possible – and preferable – to combat fraud without punishing an entire healthcare sector because of the actions of a handful of bad actors. Regrettably, it seems that the Centers for Medicare and Medicaid Services (CMS) has opted for the latter approach, despite Congress having given the agency adequate authority to drive fraudulent suppliers from the Medicare program.
The Fraud-Fighting Tools CMS Has Not Used
Section 427 of the Beneficiary Improvement and Protection Act (BIPA) of 2000 requires CMS to ensure that Medicare payments for custom fabricated orthotics and all prosthetics are furnished by “qualified practitioners” and “qualified suppliers.” The orthotics and prosthetics profession supported this effort and consistently has pushed to have this requirement implemented. Currently, 14 states have enacted orthotics and prosthetics licensure statutes. In 2005, CMS issued Transmittal 656 to Medicare payment contractors specifying that contractors must have claims processing edits in place to make sure that in those states where prosthetics or orthotics must be provided by a licensed or certified orthotist or prosthetist, payments are made only to practitioners and suppliers that meet relevant state orthotics and prosthetics licensure laws. However, CMS has not taken concrete steps to enforce this requirement. For example, in 2009, a “60 Minutes” expose demonstrated that CMS was paying unlicensed providers for orthotic and prosthetic services. The amount of Medicare funds inappropriately paid by CMS was in the tens of millions. The fraud discussed in that report involved Florida, a state with orthotics and prosthetics licensure requirements.
Since Congress passed BIPA, AOPA and its members have met with CMS administrators and staff regarding implementation of the law, and in 2007, we were told that proposed regulations would be issued by the end of that year. We are still waiting. On June 25, 2013, AOPA shared with CMS the results of two studies that demonstrate that CMS had been paying unlicensed suppliers.
- In one study, the health economics and policy consulting firm Dobson-DaVanzo examined Medicare claims data from 2007-2011 and did not find significant changes in the distribution of payments to medical supply facilities with uncertified orthotics and prosthetics professionals on their staffs. We note that orthotist and prosthetist licensing requirements in most states track very closely with the typical certification requirements of training and education so that a person who is not certified will almost never meet eligibility for licensure. It is possible to be certified and not licensed, but it is virtually impossible to be licensed and not certified.
- In the other study, conducted in 2013, orthotics and prosthetics suppliers who were receiving Medicare payments were contacted in three licensure states and asked if they had a licensed orthotics and prosthetics professional on staff. This study revealed that 65 out of 78 surveyed suppliers by their own admission did not have a licensed professional on staff.
In a letter to AOPA dated August 2, 2013, CMS Administrator Marilyn Tavenner denied that CMS has been paying unlicensed orthotics and prosthetics suppliers. In the letter, Administrator Tavenner states that systematic claims edits have been in place since 2005 to deny claims submitted by unlicensed suppliers in nine states with orthotics and prosthetics licensure requirements (AL, FL, IL, NJ, OH, OK, RI, TX, and WA) and that the agency is implementing claims edits for the remaining five states with licensure requirements (AR, GA, KY, MS and TN). (This was reported in a Medicare Learning Network Matters article on the same day.) This amounts to an admission by the agency that it has been paying unlicensed suppliers in at least five licensure states (and CMS has omitted any reference to Pennsylvania and Iowa, both of which have enacted O&P licensure as well). Also, the effectiveness of the claims edits in the other nine states is questionable, in light of the fraud that has been documented in two of these states (FL and TX) since 2005 when these edits reportedly were implemented.
It is difficult to understand how the relative proportion of Medicare payments to non-certified orthotics and prosthetics suppliers is unchanged if unlicensed providers no longer are receiving payments in states where certification is required. We have seen evidence of only a small reduction in the proportion of payments to non-certified orthotics and prosthetics personnel since 2009. This also is supported by the results of the independent survey of orthotics and prosthetics suppliers, which showed that unlicensed, non-certified suppliers continue to provide and be paid for orthotics and prosthetics furnished to Medicare beneficiaries, even in states where licensure is required.
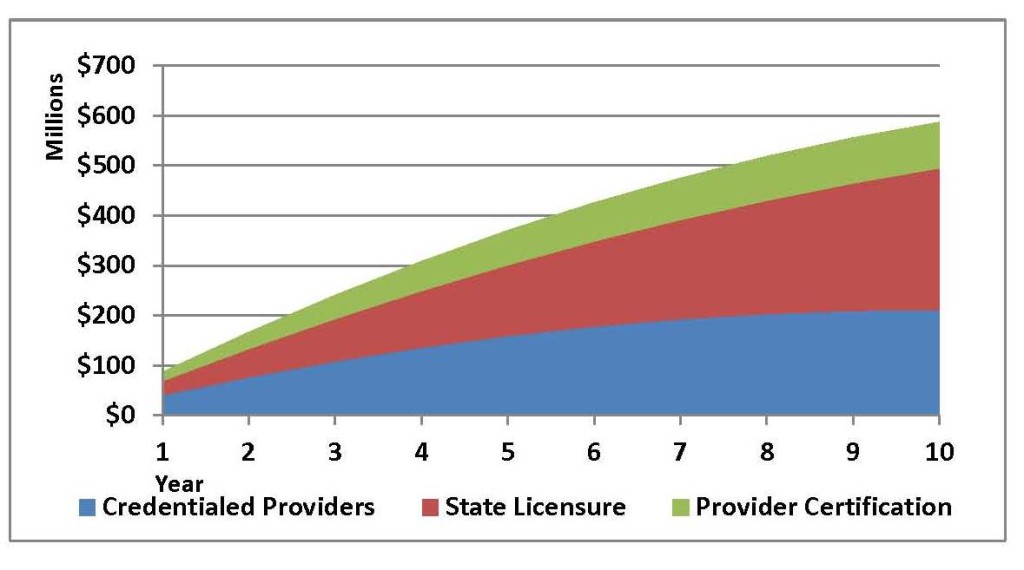
H. R. 3112, the Medicare Orthotics and Prosthetics Improvement Act, has been introduced in Congress and would build upon the fraud-fighting provisions included in BIPA. It would help reduce fraud, protect patients, and save Medicare funds by keeping out fraudulent providers in the first place. As the Dobson-DaVanzo report notes: “If CMS was to actively enforce that unlicensed providers cannot receive payment for providing orthotics and prosthetics services to Medicare beneficiaries within a licensure state, Medicare savings could be realized. Under such enforcement of limiting payments to providers with proven licensure and standards of training and experience, payments to unqualified providers would be eliminated. As the ‘60 Minutes’ special suggested, allowing non-certified personnel to provide these services, especially in states with licensure, could lead to fraud and abuse in orthotics and prosthetics services, as well as expose patients who received these services to inappropriate or substandard care. Therefore, shifting payments to only certified providers could result in better care for beneficiaries and lower Medicare payments.” An estimate of how much could be saved by implementation of these provisions is provided in the following excerpt from a 2009 report prepared by Morrison Informatics.
Table 1 – First Year Medicare Savings Estimated Range Following H.R. 2479 (now H.R. 3112) Provisions, with Amendment
| Provision | Medicare SavingsRange | Proportion of Savings |
| Credentialed Providers | $40 – $101 | 47% |
| State Licensure | $28 – $71 | 33% |
| Provider Certification | $18 – $44 | 20% |
| Total | $86 – $216 | 100% |
Figure 1 – Minimum Cumulative 10-Year Medicare Savings Projection Following H.R. 2479 (now H.R. 3112) Provisions, with Amendment
Concerns about CMS’s failure to implement BIPA Section 427 were brought to the agency’s attention most recently in a letter from the Chair of the House Ways and Means Committee and the Chair of the Health Subcommittee. CMS’s response, dated March 6, 2014, stated that CMS is developing a notice of proposed rulemaking and anticipates that it will be published in 2014. AOPA is skeptical, since we have been told by Ms. Tavenner and other CMS administrators in the past that proposed regulations were forthcoming. In its response, CMS also said that “when a state has enacted a new licensure law, CMS implements an edit that immediately limits payment to only those suppliers that have a specialty of orthotics and prosthetics on their enrollment applications. Then the [National Supplier Clearinghouse] determines whether all orthotic and prosthetic suppliers in the affected state have the required licenses or certifications.” However, the National Supplier Clearinghouse generally scrutinizes a potential orthotics and prosthetics supplier only when the supplier seeks a new Medicare provider number and on a regular three year re-enrollment cycle thereafter. AOPA is not aware of actions taken by National Supplier Clearinghouse to monitor orthotics and prosthetics suppliers for licensure after granting a Medicare number.
In summary, CMS currently has several tools at its disposal to bolster its efforts to fight fraud in the orthotics and prosthetics field, yet it has failed to avail itself of its full arsenal. It has not issued any regulations to implement Section 427 of BIPA, and edits to prevent payment to unlicensed orthotics and prosthetics suppliers have not been implemented fully. These shortcomings were highlighted by the HHS Office of Inspector General in its October, 2012 report entitled, “CMS Has Not Promulgated Regulations to Establish Payment Requirements for Prosthetics and Custom-Fabricated Orthotics,” but still no rules have been promulgated.
RAC Audits and the ALJ Appeals Backlog
Instead of using tools to keep bad actors from participating in the orthotics and prosthetics sector, CMS has ramped up the Recovery Audit Contractor (RAC) program, which has had the effect of punishing legitimate providers.
While CMS makes payments to unlicensed and unaccredited providers, contravening Congress’s intention, legitimate suppliers have been subject to RAC and prepayment audits conducted by contractors who appear to play by their own set of rules. It also appears that RAC audits penalize suppliers for paperwork or documentation errors as often, or more often, than it catches those perpetrating fraud. This sometimes results in legitimate providers, especially those who are small businesses, suffering cash flow problems or going out of business. AOPA estimates that roughly 100 orthotics and prosthetics suppliers have gone out of business, at least in part due to these audit/recoupment related cash flow problems. The impact of these closings extends beyond economics and business—it directly and negatively affects individuals with limb loss, as they have been deprived of long-standing, clinically-beneficial relationships with their health care providers. We note that AOPA has sued the U.S. Department of Health and Human Services (HHS) over RAC audits and how they are being applied to orthotics and prosthetics suppliers.
We feel that certain actions by CMS have compromised the due process rights of orthotics and prosthetics suppliers. For example, CMS issued a “Dear Physician” letter on its website in August, 2011 that had the effect of establishing new policy for payment for artificial limbs, and it applied the new policy retroactively in RAC and prepayment audits as to claims for dates of service as much as two years before the policy was issued in the letter.
There has been an explosion in the number of RAC audit claims under Medicare Part B for artificial limbs that are appealed to the Administrative Law Judge (ALJ) level. Congress and CMS have provided some modest relief for Medicare Part A providers, but none of this relief has been extended to Part B claims for artificial limbs. While we appreciate the difficult task facing the Office of Medicare Hearings and Appeals (OMHA), timely redress of improperly denied payments is critical. Many suppliers, particularly in the orthotics and prosthetics field, are small businesses that do not have the luxury of waiting months for payment of services legitimately furnished. In fact, just last year, 35 Members of Congress wrote to HHS Secretary Kathleen Sebelius that well-intentioned efforts to reduce fraud and abuse in Medicare may be harming access for vulnerable Medicare beneficiaries and placing undue burdens on legitimate orthotic and prosthetic providers. In a context of increasingly aggressive CMS audits, OMHA’s decision to suspend ALJ review of provider and supplier claims is devastating to suppliers who deliver Medicare services to over 40 million beneficiaries.
Congress showed that it understood the importance of timely processing of Medicare appeals when it included in BIPA a requirement that an ALJ issue a decision about a case within 90 days of the date when the appeal request was filed. However, by OMHA’s own admission, the current wait time for a hearing before an ALJ has increased to 16 months. In some areas that wait is as long as 26 months, which is unacceptable.
At the February 12, 2014 OMHA public hearing on this issue, Judge Griswold gave an explanation of OMHA’s position, but offered few if any short-term remedies that would restore the right of a timely ALJ hearing to providers. With ALJs siding fully with appellants in over half of all decisions, ALJ hearings realistically amount to a provider’s primary means of challenging costly and often prejudicial CMS auditor decisions. As OMHA is leaving Medicare providers without an avenue of redress against auditors’ payment denials, we believe it is only fair that CMS suspend these audits until an appropriate, timely, and statutorily required system providing due process to providers is restored.
Conclusion
In conclusion, AOPA wants to continue to work with Congress and CMS to ensure that those who prey on Medicare beneficiaries do not find the orthotics and prosthetics sector an easy place to establish and operate a fraud scheme. We offer our support for developing more effective means to fight Medicare fraud that does not punish legitimate suppliers who are playing by the rules. We believe that the fairest and most effective system is one that prevents fraud before it starts, and we hope that Congress will direct CMS to develop a system taking the pathways outlined in both Section 427 of BIPA 2000 and H.R. 3112 to deter fraud, promote program integrity, and protect the due process rights of legitimate orthotics and prosthetics suppliers.
AOPA appreciates the efforts of the Chairman of the Committee and of the Subcommittee on Health for working with us to find ways to better regulate our profession. We hope to continue to work with you to improve the quality of care we deliver to patients who need orthotics and prosthetics and to protect the integrity of the Medicare program.